Primary Breast Sarcoma
Abstract
Introduction
Primary sarcomas of the breast are <0.1% of all malignant tumours of the breast. To date, there are 13 major breast sarcoma series in English literature. This study adds to these series characterizing primary breast sarcoma among Philippine patients.
Methods
All breast biopsies from the pathology records of the University of the Philippines-Philippine General Hospital (UP-PGH) were searched for breast sarcoma cases from January 2000 to December 2010. Metaplastic carcinomas and phyllodes tumors were excluded.
Results
There were 52 patients (45 female, 7 male) ranging in age 25-83 years (median 46 years). Majority had lump, ten cases with pain. No history of previous cancer was given. No history of prior radiation was found. Histopathological diagnoses were spindle cell sarcoma (n=13), fibrosarcoma (n=6), liposarcoma (n=6), MPNST (n=5), stromal sarcoma (n=5), angiosarcoma (n=4), MFH (n=4), leiomyosarcoma (n=3), rhabdomyosarcoma (n=3), chondrosarcoma (n=2), and synovial sarcoma (n=1). Tumors were with grade 1 (n=18), grade 2 (n=8), and grade 3 (n=10). Necrosis was noted in 6 cases. Simple mastectomy was done in 19 cases (37%), MRM in 31 cases (59%), while 2 far advanced had no surgery (3%). None had adjuvant radiotherapy or chemotherapy. The duration of follow-up for 45 patients ranged from 1 – 117 months, excluding those who were lost to follow-up. All 15 deaths were due to progressive disease. Recurrences were observed in 9 patients. The disease-free survival (DFS) and overall survival (OS) was 73%and 75%, respectively. On multivariate analysis, DFS and OS were significantly correlated with size (HR=113.63; p=0.019 and HR=77.36; p=0.037), grade (HR=20.73 ; p=0.003andHR= 39.57; p= 0.004), and having a histology of angiosarcoma (HR=35.20 ; p=0.005and HR= 50.74; p=0.007), respectively.
Conclusion
Sarcoma remains an important clinical entity among primary breast cancers.
Author Contributions
Academic Editor: Anubha Bajaj, Consultant Histopathology, A.B. Diagnostics, New Delhi, India.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2022 Mary Ondinee Manalo Igot, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Primary breast sarcoma (PBS) is a malignant tumor arising from the mesenchymal tissue of the mammary gland.1, 2, 3As of the present, there is no consensus according to the exact definition of breast sarcoma. Some authors excluded cystosarcoma phyllodes from their studies due to its epithelial components, but others do not distinguish between other subtypes of breast sarcoma and cystosarcoma because of similar survival and clinical course.
PBS is an extremely rare and heterogeneous disease initially described in 1887.10No group of investigators has had substantial personal experience encompassing much of the clinicopathological spectrum of this disease.3The true incidence of this malignancy is still unknown. One study reported that this occurs with an approximate incidence of 17 new cases per million women.4In another series, the annual incidence rate is 44.8 new cases per ten million women. At the Mayo Clinic, all cases pathologically diagnosed in their institution from 1910 to 2000 were reviewed and they reported that primary breast sarcomas account for 0.0006% of all their breast malignancies.5
Because this tumor is rare, the majority of publications are case reports or small series. This study contributes to the literature on this disease and characterizes the Filipino patient with primary breast sarcoma, excluding cystosarcoma phyllodes.
Methods
This retrospective cohort included all cases diagnosed pathologically with primary breast sarcoma at the University of the Philippines-Philippine General Hospital (UP-PGH) Department of Pathology from January 2000 to December 2010. Excluded were patients aged <19 years, and those with other primary or metastatic disease to the breast, carcinosarcoma, spindle cell carcinoma, metaplastic carcinoma, or cystosarcoma phyllodes.
Clinical charts and surgical notes were retrospectively reviewed and the following information were collected: age, gender, size of tumor, clinical presentation, duration of symptoms, history of radiation, type of surgery, local recurrences, systemic metastases, and survival data. Follow-up information was obtained from patient records, death certificates, and correspondence. The patients were searched actively from the three medical records section (Main Records Section, Medical Oncology Records Section and Surgical Oncology Records Section) of the UP-PGH. Addresses and contact numbers were noted and correspondence was attempted via phone calls and letters. For those patients with vague addresses, attempts were made to contact their local government to help the authors find the patients for more accurate survival data.
Patient characteristics, method of biopsy, type and extent of surgery, histopathology, forms of treatment given, and sites of metastases were determined.
Continuous variables were summarized as means, medians, and ranges while categorical variables presented as frequencies and proportions. Stata Statistics Data Analysis software version 12.0 was used to analyze data. Three years OS and DFS following diagnosis were estimated based on the Kaplan-Meier method. Associations between age, size, grade, margin status, surgery and survival were evaluated based on fitting Cox proportional hazards models. All calculated p-values were two-sided and p-values <0.05 were considered statistically significant.
A pre-approved protocol submitted to the Research Ethics Board (UPMREB) Review Panel was used in this study.
Results
The histopathologic diagnoses of all breast biopsies sent to the Department of Pathology (Figure 1) and the relative frequency of primary breast sarcomas (Figure 2) are shown below. PBS account for 0.12-0.69% of all breast masses and 0.17-0.99% of all malignancies submitted for histopathologic analysis.
Table 1 profiles the 52 patients with PBS retrieved from 2000 to 2010 (Figure 2). Five patients did not follow-up and could not be located after surgery; two patients who were metastatic and symptomatic at onset did not undergo any surgery. These seven patients were excluded from the analysis. A total of 45 patients were included in the survival data and association of risk factors.
Table 1. Primary breast sarcoma, January 2000 to December 2010, UP-PGH| Characteristics | No. of Patients (n=52) | % |
| Age, years | ||
| Median | 46 | |
| Range | 25-83 | |
| Size, cm | ||
| Median | 7.4 cm | |
| Range | ||
| < 5cm | 12 | 23 |
| 5-10cm | 21 | 41 |
| > 10cm | 19 | 36 |
| Gender | ||
| Male | 7 | 13 |
| Female | 45 | 87 |
| M/F Ratio | 1:0.15 | |
| Histologic types | ||
| Spindle-cell sarcoma | 13 | 26 |
| Fibrosarcoma | 6 | 12 |
| Liposarcoma | 6 | 12 |
| MPNST | 5 | 10 |
| Stromal sarcoma | 5 | 10 |
| Angiosarcoma | 4 | 7 |
| MFH | 4 | 7 |
| Leiomyosarcoma | 3 | 5 |
| Rhabdomyosarcoma | 3 | 5 |
| Chondrosarcoma | 2 | 4 |
| Synovial sarcoma | 1 | 2 |
| Tumor grade | ||
| Grade 1 | 18 | 34 |
| Grade 2 | 8 | 15 |
| Grade 3 | 16 | 31 |
| Not graded | 10 | 20 |
| Initial treatment | ||
| Simple Mastectomy Alone | 19 | 37 |
| Modified Radical Mastectomy | 31 | 59 |
| Plus adjuvant CT | None | None |
| Palliative RT | None | None |
| Palliative RT + CT | None | None |
Figure 1.Histopathologic diagnoses of breast biopsies by the Department of Pathology
Figure 2.Histopathologic diagnoses of all the breast cancers by the Department of Pathology, UP-PGH, January 2000 – December 2010
The 45 women had age range 25-83 (mean 46 years, median 46). There were 7 male patients with age range 32-64 (mean 41 years, median 51). Majority had with a lump, ten cases had pain. One case presented as an incidental mammographic finding. No history of previous cancer was elicited in any of the patients. No history of prior radiation was found in any case.
None underwent radiotherapy or chemotherapy.
Gross pathological description was available for 15 cases. Seven tumors were described as well-circumscribed, eight as infiltrative. Histopathological diagnoses were spindle cell sarcoma (n=13), fibrosarcoma (n=6), liposarcoma (n=6), MPNST (n=5), stromal sarcoma (n=5), angiosarcoma (n=4), MFH (n=4), leiomyosarcoma (n=3), rhabdomyosarcoma (n=3), chondrosarcoma (n=2), and synovial sarcoma (n=1). Tumors were graded as grade 1 (n=18), grade 2 (n=8), and grade 3 (n=10). Necrosis was noted in 6 cases.
Simple mastectomy was done in 19 cases (37%), MRM in 31 cases (59%), while 2 far-advanced had no surgery (3%). One patient initially underwent wide excision but the margins were positive, so she had simple mastectomy subsequently with clean margins. Of the 19 patients who underwent simple mastectomy, all had clear margins. Majority of the tumors (58%) subjected to simple mastectomy were <5 cm. Of the 31 patients who underwent MRM, three had lymph node involvement and positive margins. Those with no available margin status in their pathology report were assumed to have negative margins.
Forty-five patients had follow-up data ranging from 1-117 months; the others were lost to follow-up. There were fifteen deaths, all tumor-related. All patients with initially unresectable tumors died of their disease. Recurrences were observed in 9 patients; one chest wall recurrence occurred after mastectomy. Distant metastases developed subsequently in 8 patients. Four patients metastatic on initial presentation died within less than a year.
The 1-, 2-, and 3-year overall survival (OS) for the whole population was 89%, 81% and 75%, respectively. Disease-free survival rates at 1, 2, and 3 years were 78 %, 73 %, and 73% respectively (Figure 3, Figure 4). Also shown are the survival curves comparing the effect of tumor size, grade and histology (Figure 5, Figure 6, Figure 7). 6.38 patients will recur per 1000 person-months, while 5.85 patients will die per 1000 person-months.
Figure 3.Overall survival of patients with primary breast sarcoma in the Philippine General Hospital from 2000-2010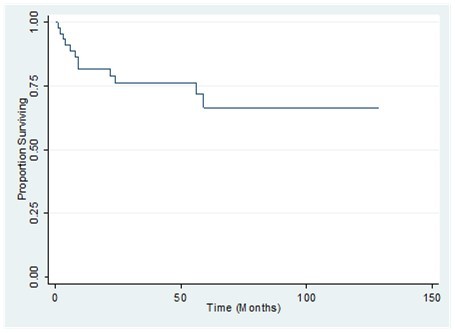
Figure 4.Disease-free survival of patients with primary breast sarcoma in the Philippine General Hospital from 2000-2010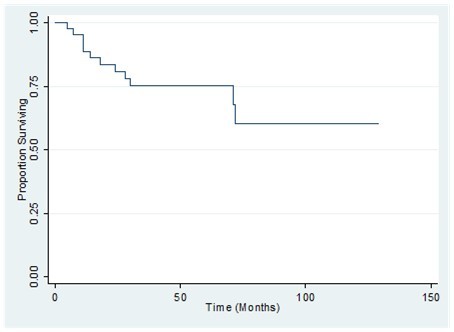
Figure 5.Comparison of the overall survival by tumor size of patients with primary breast sarcoma in the Philippine General Hospital from 2000-2010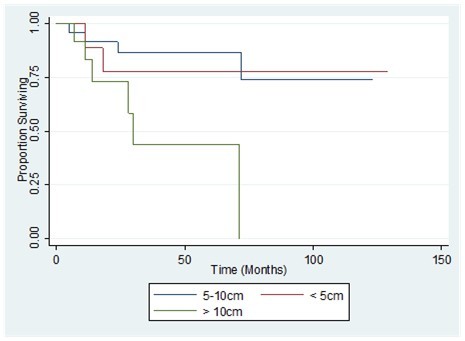
Figure 6.Comparison of the overall survival by tumor grade of patients with primary breast sarcoma in the Philippine General Hospital from 2000-2010
Figure 7.Comparison overall survival by tumor histology of patients with primary breast sarcoma in the Philippine General Hospital from 2000-2010
Prognostic factors for disease recurrence are shown in Table 2. On univariate analysis, tumor grade (HR=10.48; p=0.001) and having the histology of angiosarcoma (HR=13.47; p=<0.001) were significantly correlated with recurrence. On multivariate analysis, tumor size (HR=113.63; p=0.019), grade (HR=20.73; p=0.003), and having the histology of angiosarcoma (HR=35.20; p=0.005) were all significantly correlated with recurrence, while controlling for the confounding effect of clinically important factors such as age, margin status, and type of surgery.
Prognostic factors for mortality are shown in Table 3. On univariate analysis, tumor grade (HR=18.64; p=0.001) and having the histology of angiosarcoma (HR=11.03; p=<0.001) were significantly correlated with mortality. On multivariate analysis, tumor size (HR=77.36; p=0.037), grade (HR=39.57; p=0.004), and having the histology of angiosarcoma (HR=50.74; p=0.007) were all significantly correlated with mortality, while controlling for the confounding effect of clinically important factors such as age, margin status, and type of surgery.
Table 2. Prognostic Factors for Disease Recurrence from Primary Breast Sarcoma
| Disease Free Survival (%) | Univariate Results | Multivariate Results | |||
| HR | p | HR | p | ||
| Tumor Size | |||||
| <5 cm | 78 | 1.00 | - | 1.00 | - |
| 5-10 cm | 86 | 0.75 | 0.783 | 17.07 | 0.100 |
| >10 cm | 49 | 3.20 | 0.158 | 113.63 | 0.019 |
| Grade | |||||
| 1 | 91 | 1.00 | - | 1.00 | - |
| 2 | 88 | 2.41 | 0.336 | 2.71 | 0.374 |
| 3 | 40 | 10.48 | 0.001 | 20.73 | 0.003 |
| Margin Status | |||||
| - | 77 | 1.00 | - | 1.00 | - |
| + | 67 | 1.81 | 0.575 | 1.04 | 0.972 |
| Type of Surgery | |||||
| SM | 75 | 1.00 | - | 1.00 | - |
| MRM | 77 | 1.10 | 0.866 | 0.14 | 0.067 |
| Age | |||||
| >40 | 80 | 1.00 | - | 1.00 | - |
| ≤40 | 66 | 1.76 | 0.366 | 0.76 | 0.772 |
| Histology | |||||
| Others | 84 | 1.00 | - | 1.00 | - |
| Angiosarcoma | 0 | 13.47 | <0.001 | 35.20 | 0.005 |
Table 3. Prognostic Factors for Mortality from Primary Breast Sarcoma
| Overall Survival (%) | Univariate Results | Multivariate Results | |||
| HR | p | HR | p | ||
| Tumor Size | |||||
| <5 cm | 78 | 1.00 | - | 1.00 | - |
| 5-10 cm | 86 | 0.73 | 0.715 | 23.86 | 0.122 |
| >10 cm | 44 | 3.65 | 0.122 | 77.36 | 0.037 |
| Grade | |||||
| 1 | 91 | 1.00 | - | 1.00 | - |
| 2 | 88 | 4.03 | 0.097 | 7.19 | 0.139 |
| 3 | 32 | 18.64 | 0.001 | 39.57 | 0.004 |
| Margin Status | |||||
| - | 77 | 1.00 | - | 1.00 | - |
| + | 50 | 1.49 | 0.704 | 0.60 | 0.678 |
| Type of Surgery | |||||
| SM | 74 | - | - | - | - |
| MRM | 77 | 1.08 | 0.901 | 0.32 | 0.207 |
| Age | |||||
| >40 | 79 | - | - | - | - |
| ≤40 | 63 | 1.71 | 0.387 | 0.61 | 0.613 |
| Histology | |||||
| Others | 84 | - | - | - | - |
| Angiosarcoma | 0 | 11.03 | <0.001 | 50.74 | 0.007 |
Table 4. Major breast sarcoma series in the English literature
| Author | N cases/period | Median age(years) | Median size (cm) | Top Diagnosis | PrognosticFactors |
| Berg et al(1962) | 25/- | 48 | 6.0 | Liposarcoma, fibrosarcoma | Positive margins |
| Oberman(1965) | 13/30 yrs | 56 | 7.1 | Fibrosarcoma, rhabdomyosarcoma, desmoid | Size, type of surgery |
| Norris & Taylor(1968) | 32/- | 49 | 4.0 | Osteosarcoma, MFH, leiomyosarcoma | Size, contour, atypia, mitotic index |
| Barnes &Pietruszka (1977) | 10/31 yrs | 51 | 6.3 | Fibrosarcoma, rhabdomyosarcoma. leiomyosarcoma | Tumour contour, atypia, mitosis |
| Callery et al(1985) | 25/33 yrs | 54 | 4.0 | Fibrosarcoma, MFH, Leiomyosarcoma | - |
| Stanley et al (1988) | 4/- | 61 | - | MFH, angiosarcoma | - |
| Pollard et al(1990) | 25/81 yrs | 55.4 | 5.9 | MFH, leiomyosarcoma, fibrosarcoma | Type of surgery |
| Johnstone et al(1993) | 10/12 yrs | 28 | - | Angiosarcoma, MFH, rhabdomyosarcoma | - |
| Smola et al(1993) | 8/23 yrs | 56 | 12.8 | Chondrosarcoma, MFH, leiomyosarcoma | - |
| Gutman et al(1994) | 60/51 yrs | 48 | 6.5 | Angiosarcoma, spindle cell sarcoma | Size, multifocal lesions, vascular/lymphatic/chest wall invasion |
| North et al(1998) | 25/31 yrs | 55 | 6.0 | Angiosarcoma, spindle cell sarcoma, fibrosarcoma | Type of surgery |
| Barrow et al(1999) | 59/43 yrs | 45 | - | Fibrosarcoma, angiosarcoma, osteosarcoma | Size, margin status, histology |
| Zelek et al(2003) | 83/37 yrs | 47 | 6.5 | MFH, angiosarcoma, leiomyosarcoma | Grade, size |
| Adem et al(2004) | 42/80 yrs | 40 | 5.7 | Fibrosarcoma, angiosarcoma, leiomyosarcoma |
Discussion & Literature Review
Etiology
The risk of developing breast sarcoma is largely unknown. Some authors showed a significant correlation between external beam radiation of the breast or chest wall and sarcoma.28Another study demonstrated a larger percentage of angiosarcomas as a histopathological subtype post-irradiation therapy sarcoma when compared with other subtypes of sarcoma. The latent time interval between radiotherapy and breast sarcoma was 12 years and 8 months. For breast angiosarcoma this period of time was even shorter with only 8 years and 8 months.7 Other studies had comparable results.7 Another proposed risk factor, especially for angiosarcomas, is a chronic lymphedema of the breast and the arm.38 In the future, the number of sarcomas may grow because the increasing numbers of patients undergoing radiotherapy. This UP-PGH study had no patients with prior exposure to radiation or chronic lymphedema.
Clinical Presentation
Breast sarcoma is mostly diagnosed in patients who are in their 5th or 6th decade of life, and the UP-PGH data is comparable to the largest review done by Al-Benna42. In that review, the weighted mean age upon diagnosis was 50.0 years, with 98.5% occurring in women and 1.5% in men. The mean tumor size was between 4.8- 6.6 cm (range: 0.8 cm-40cm) in most studies. McGregor26 reported median tumor size of 3 cm and Adem43 had a mean tumor size of 10 cm for angiosarcomas. In contrast to the UP-PGH study the mean age of diagnosis is 46 years, with 87% occurring in women and 13% among men and the median tumor size is 7.4 cm. It has been postulated that the menopausal status influences breast sarcoma development, but there is little evidence to support this. The typical patient with a breast sarcoma has a unilateral, well-defined, mobile and painless breast lump which often grows more rapidly than epithelial breast carcinoma
Histological Diagnosis
Primary breast sarcomas are as histologically heterogenous as non-breast tissue sarcomas.39 It needs a thorough histologic review by a qualified and experienced soft tissue pathologist.40A subtype evaluation in the breast is limited due to its rarity and at times, the heterogeneity that these tumors present with may lead to misclassification.28 There is debate on the exclusion of cystosarcoma phyllodes from PBS studies. Some authors excluded the former because these tumors have a benign epithelial component in addition to a malignant mesenchymal component. However, some classify malignant phyllodes as a subtype of PBS due to its similar clinical course. PBS on the other hand, arises from the mesenchymal tissue of the mammary gland, and the pathologic definition of this entity is clear.11
Also, PBS should be distinguished from metaplastic carcinomas (carcinosarcomas) of the breast, which represent as various combinations of poorly differentiated ductal carcinoma with mesenchymal and squamous components. There are clinicians in literature who classify and manage them as a variant of ductal adenocarcinomas of the breast. Therefore, when facing a spindle cell neoplasm in an epithelial organ such as the breast, one should be careful in rendering the diagnosis of sarcoma. In this setting, immunohistochemistry using the right antibodies is a major contribution. Berg defined stromal sarcomas of the breast in 1962 as a group of mesenchymal malignant tumors with fibrous, myxoid and adipose components, excluding malignant cystosarcoma phyllodes and lymphomas.12
However, series in the literature have included many different entities under the rubric of sarcomas such as cystosarcoma phyllodes andlymphosarcoma. 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28For this study, we chose to categorize primary breast sarcomas in histogenic terms, similar to other soft-tissue sarcomas, thus including angiosarcomas, and excluding malignant cystosarcomas phyllodes and carcinosarcoma (Table 4).1, 2, 3, 12, 29, 30, 31
In this UP-PGH data, the most common histologies were spindle cell sarcoma, fibrosarcoma, and liposarcoma constituting 50% of the whole population. This was comparable with the study done by Berg12 and by North44.
The current 2010 American Joint Committee on Cancer TNM system45 incorporates four major changes compared with the 2002 system, two of the changes pertain to histology. First, it excludes four histologic types: gastrointestinal stromal tumor (GIST), desmoid tumor, Kaposi sarcoma, and infantile fibrosarcoma. Second, it adds the following histologic types: angiosarcoma, extra skeletal Ewing sarcoma, and dermatofibrosarcomaprotuberans. For the purposes of this study, re-classification of histologic subtypes before the changes were made was not done because of no data. The report of the pathologist was taken as it was and everything written in the report was recorded in the data collection sheet as it was.
Patterns of Metastasis
The results from this UP-PGH study showed that the lung was the most common site of distant metastases (75%), followed by the liver (25%).
Metastases from breast sarcoma spread hematogenously, and typically to the lungs, bones, and liver. Lymph node metastases are rare. In most cases of lymph node metastasis, the histopathologic subtype carcinosarcoma should be ruled out. Breast skin and the nipple areola complex are rarely involved by breast sarcomas, but angiosarcomas may be associated with a bluish discoloration of the skin overlying the lesion.7,8,15
Treatment of Breast Sarcomas
There is still no definitive consensus regarding the treatment of PBS, even if simple mastectomy without axillary dissection is still widely regarded as the gold standard,11the only potentially curative modality for breast sarcoma. There are major variations in the extent of local therapy, ranging from wide local excision30, 32to radical mastectomy.33, 34 Recourse to chemotherapy and radiotherapy has also differed from one center to another 29, 31, 32A key article related to soft tissue sarcomas was the collaborative meta-analysis published in 1997, which showed that adjuvant chemotherapy with doxorubicin-based regimens yielded a statistically significant improvement in disease-free survival but without a considerable impact on overall survival.35 Moreover, a recent randomized trial comparing intensive adjuvant epirubicin plus ifosfamide versus no treatment showed, despite its small size, a significant improvement in both disease-free and overall survival.36 Adjuvant radiotherapy may improve locoregional control but its impact on survival is still unclear.
According to the clinicopathologic series done at the Mayo Clinic, lymphatic spread is uncommon as shown by the absence of axillary lymph node metastasis in their cases, and therefore axillary node dissection is not necessary. When lymph node metastasis is present, the diagnosis of a metaplastic carcinoma should be considered even in the presence of a pure spindle cell neoplasm, or that the sarcoma is far advanced.5
Prognostic Factors of Patients with Breast Sarcomas
Tumor size seems to be the most frequently reliable prognostic factor in many of these series, as in breast carcinomas and soft tissue sarcomas. 1, 30, 31, 33 Other reported prognostic factors are the histopathological diagnosis,31infiltrative features,2histopathologic grading,2,30,31presence of positive margins,12,31and extent of surgery for local recurrence.12,31 Some authors also found age to be of prognostic importance.19Angiosarcoma was associated with poor outcomes in some series.1,2,30,31Margin status is a major risk factor for recurrence as it occurs in any neoplastic entity, and some authors advised adjuvant radiotherapy for cases with positive margins,3 or less than 2 cm of clear margins.28In this UP-PGH study, tumor size >10 cm, positive margins, tumor grades 2-3, and a histologic diagnosis of angiosarcoma are well correlated with lower DFS and OS, comparable with similar studies.
Summary & Reflections
Breast sarcomas are primarily non-epithelial malignancies that arise from the connective tissue within the breast. They can be primary or secondary, as a result of treatment (usually RT) from previous breast malignancies. They are histologically heterogeneous, thus definitive histologic diagnosis maybe difficult if not seen by an experienced pathologist. They are extremely aggressive and once not properly diagnosed or managed early, have poor prognosis. Due to its rarity, it is difficult to conduct a prospective study to establish a standard of care for patients with primary breast sarcoma.
This UP-PGH series is similar to other series done in US and Europe, with findings as follows: (1) the larger the size, the poorer the prognosis; (2) the higher the grade, the poorer the prognosis; (3) angiosarcoma as a histologic subtype is associated with poor outcomes; and (4) the lung and liver are the most common sites of metastases. These are not different from the natural behavior of soft tissue sarcomas from other parts of the body. Therefore, primary breast sarcomas should be managed as such.
In the diagnosis of primary breast sarcoma, core needle biopsy is the procedure of choice for definitive diagnosis. Appropriate diagnostics should be ordered for accurate staging. According to the NCCN guidelines, chest CT scan should be ordered for all and abdominopelvic CT scans should be considered for angiosarcoma. For angiosarcomas, given the possible catastrophic outcomes, they should be offered early on maximal treatment, and that is, surgery, chemotherapy, and radiotherapy.
Due to the rarity of this disease, a multidisciplinary approach is recommended to properly manage this particular subset of sarcomas. Histologic review by an experienced soft tissue pathologistis critical in making the diagnosis and determining the histologic subtype. If diagnosis is still doubtful, appropriate immunostaining should be applied.
References
- 2.Barnes L, Pietruszka. (1977) Sarcomas of the breast: a clinicopathological analysis of ten cases. Cancer. 40, 1577-1585.
- 3.Callery C D, Rosen P P, Kinne D W. (1985) Sarcoma of the breast: a study of 32 patients with reappraisal of classification and therapy. Ann Surg. 201, 527-532.
- 5.Adel C, Reynolds C, Ingle J N, Nascimento A G. (2004) Primary breast sarcoma: Clinicopathological series from the Mayo Clinic and review of literature. , British Journal of Cancer 91(2), 237-241.
- 6.The Philippine General. Hospital. http://www.scribd.com/doc/11287963/The-Philippine-General-Hospital
- 7.Blanchard D K, Reynolds C A, Grant C S, Donohue J H. (2003) Primary nonphylloides breast sarcomas. , The American Journal of Surgery 186(4), 359-361.
- 8.Bousquet G, Confavreux C, Magne N. (2007) Outcome and prognostic factors in breast sarcoma: a multicenter study from the rare cancer network. Radiotherapy & Oncology. 85(3), 355-361.
- 9.Fields R C, Aft R L, Giliaders W F, Eberlain T J, Margenthaler J A. (2008) Treatment and outcomes of patients with primary breast sarcoma. , The American Journal of Surgery 196(4), 559-561.
- 10.Jardines L. (2000) Other cancers in the breast. In. et al (eds): Diseases of the Breast , Philadelphia, PA, Lippincott-Raven 876-882.
- 11.JR Petrek JA Phyllodes tumors In Harris, Lippman S, Murrow M. (2000) . Diseases of the Breast , Philadelphia, PA, Lippincott-Raven 863-869.
- 12.Berg J W, DeCrosse J J, Fracchia A A, Farrow J. (1962) . , Stromal sarcomas of the breast. Cancer 15, 418-424.
- 13.Botham R J, McDonald J R, Clagett O T. (1958) Sarcoma of the mammary gland. Surg Gynecol Obstet. 107, 55-61.
- 17.Rissanen P M, Holsti P. (1968) A retrospective study of sarcoma of the breast and the results of treatment. , Oncology 22, 258-268.
- 19.Ludgate C M, Anderson T J, Langlands A O. (1977) Sarcoma of the female breast – report of a series of 30 cases. , Clin Onco 3, 97-105.
- 20.Khanna S, Gupta S, Khanna N N. (1981) Sarcomas of the breast: homogenous or heterogenous?. , J Surg Oncol 18, 119-128.
- 21.Christensen L, Schiodt T, Blichert-Toft M, Hansen J P, Hansen O H. (1988) Sarcomas of the breast: a clinico-pathological study of 67 patients with long term follow-up. Eur J Surg Oncol. 14-241.
- 22.Terrier P, Terrier-Lacombe M J, Mouriesse H. (1989) Primary breast sarcoma: a review of 33 cases with immunohistochemistry and prognostic factors. Breast Cancer Res Treat. 13, 39-48.
- 23.Pitts W C, Rojas V A, Gaffey M J. (1991) Carcinomas with metaplasia and sarcomas of the breast. , Am J Clin Pathol 95, 623-632.
- 24.Ciatto S, Bonardi R, Cataliotti L, Cardona G. (1992) Sarcomas of the breast: a multicenter series of 70 cases. , Neoplasma 39, 375-379.
- 25.Luna Vega AR, Vetto J T, Kinne D W. (1992) Primary sarcomas of the breast in women under 20 years of age. , N Y State J Med 92, 497-498.
- 26.McGregor G I, Knowling M A, Este F A. (1994) Sarcoma and cystosarcoma phyllodes tumors of the breast – a retrospective review of 58 cases. , Am J Surg 167, 477-480.
- 28.McGowan T S, Cummings B J, O’Sullivan B. (2000) An analysis of 78 breast sarcoma patients without distant metastases at presentation. Int J Radiat Oncol Biol Phys. 46, 383-390.
- 29.PAS Johnstone, Pierce L J, Merino M J. (1993) Primary soft tissue sarcomas of the breast: Local-regional control with post-operative radiotherapy. Int J Radiat Oncol Biol Phys. 27, 671-675.
- 30.Gutman H, Polock R E, Ross M I. (1994) Sarcoma of the breast: Implications for extent of therapy: The M.D. Anderson experience. , Surgery 116, 505-509.
- 31.Barrow B J, Janjan N A, Gutman H. (1999) Role of radiotherapy in sarcoma of the breast: A retrospective review of the M.D. Anderson experience. Radiother Oncol. 52, 173-178.
- 32.Chaney A W, Pollack A, McNeese M D. (2000) Primary treatment of cystosarcoma phyllodes of the breast. , Cancer 89, 1502-1511.
- 33.Henry J, Norris M D, Herbert B. (1968) Sarcoma and related mesenchymal tumors of the breast. , Cancer 22, 22-28.
- 34.Reinfuss M, Mitus J, Smolak K. (1993) Malignant phyllodes tumours of the breast: A clinical and pathological analysis of 55 cases. , Eur J Cancer 29, 1252-1256.
- 35. (1997) Sarcoma Meta-Analysis Collaboration: Adjuvant chemotherapy for localized resectable soft-tissue sarcoma of adults: Meta-analysis of individual data. , Lancet 350, 1647-1654.
- 36.Frustaci S, Gherlinzoni F, A De Paoli. (2001) Adjuvant chemotherapy for adult soft tissue sarcomas of the extremities and girdles: Results of the Italian randomized cooperative trial. J Clin Oncol. 19, 1238-1247.
- 37.Lucas J, Uy G. (2012) Five-year Clinicopathologic Review of Phyllodes Tumors of the Breast in the Philippine General Hospital 2005-2009. Division of Surgical Oncology, Head and Neck, Breast and Soft Tissue-UP-Philippine General Hospital.
- 38.May D S, Stroup N E. (1991) The incidence rate of sarcomas of the breast among women in the United States, 1973-1986. Plast Reconst Surg. 87(1), 193-194.
- 39.Chugh R, Sabel M, Feng M. (2012) Breast sarcoma: Epidemiology, risk factors, clinical presentation, diagnosis and staging.
- 40.Presant C A, Russell W O, Alexander R W. (1986) Soft-tissue and bone sarcoma histopathology peer review: the frequency of disagreement in diagnosis and the need for second pathology opinions. The Southeastern Cancer Study Group experience. , J ClinOncol 4(11), 1658-61.
- 41.Zelek L, Llombart-Cussac A, Terrier P, Pivot X, Guinebretiere J M. (2003) Prognostic factors in primary breast sarcomas: a series of patients with long term follow-up. , Journal of Clinical Oncology 21(13), 2583-2588.
- 42.Al-Benna S, Poggemann K, Steinau H U, Steinstraesser L. (2010) Diagnosis and management of primary breast sarcoma. , Breast Cancer Res Treat 122(3), 619-26.
- 43.Adem C, Reynolds C, Ingle J N, Nascimento A G. (2004) Primary breast sarcoma: clinicopathologic series from the Mayo Clinic and review of the literature. , Br J Cancer 91(2), 237-41.
